Bn molecular orbital diagram
This is the general MO diagram you need to fill with the valence electrons of BN. Boron has 3 valence electronsand nitrogen has 5 valence electrons, this makes 8 electrons.
Skip to main content. Table of contents. Intro to General Chemistry 3h 53m. Classification of Matter. Chemical Properties. Physical Properties. Intensive vs.
Bn molecular orbital diagram
None of the approaches we have described so far can adequately explain why some compounds are colored and others are not, why some substances with unpaired electrons are stable, and why others are effective semiconductors. These approaches also cannot describe the nature of resonance. Such limitations led to the development of a new approach to bonding in which electrons are not viewed as being localized between the nuclei of bonded atoms but are instead delocalized throughout the entire molecule. Just as with the valence bond theory, the approach we are about to discuss is based on a quantum mechanical model. In Chapter 2 , we described the electrons in isolated atoms as having certain spatial distributions, called orbitals , each with a particular orbital energy. Just as the positions and energies of electrons in atoms can be described in terms of atomic orbitals AOs , the positions and energies of electrons in molecules can be described in terms of molecular orbitals MOs A particular spatial distribution of electrons in a molecule that is associated with a particular orbital energy. As the name suggests, molecular orbitals are not localized on a single atom but extend over the entire molecule. Consequently, the molecular orbital approach, called molecular orbital theory A delocalized bonding model in which molecular orbitals are created from the linear combination of atomic orbitals LCAOs , is a delocalized approach to bonding. Molecular orbital theory is a delocalized bonding approach that explains the colors of compounds, their stability, and resonance. Although the molecular orbital theory is computationally demanding, the principles on which it is based are similar to those we used to determine electron configurations for atoms. The key difference is that in molecular orbitals, the electrons are allowed to interact with more than one atomic nucleus at a time. Just as with atomic orbitals, we create an energy-level diagram by listing the molecular orbitals in order of increasing energy. We then fill the orbitals with the required number of valence electrons according to the Pauli principle. This means that each molecular orbital can accommodate a maximum of two electrons with opposite spins.
Main Group Elements: Periodic Trends. How does Charle's law relate to breathing?
.
This is the general MO diagram you need to fill with the valence electrons of BN. Boron has 3 valence electrons , and nitrogen has 5 valence electrons, this makes 8 electrons. You have to start filling the orbitals from those with lowest energy to those with higher energy. In this case, you need to follow Hund's rule, which states that if two or more orbitals of equal energy are available, electrons will occupy them singly before filling them in pairs. So you end up with 2 unpaired electrons, and paramagnetism of the molecule is explained.
Bn molecular orbital diagram
None of the approaches we have described so far can adequately explain why some compounds are colored and others are not, why some substances with unpaired electrons are stable, and why others are effective semiconductors. These approaches also cannot describe the nature of resonance. Such limitations led to the development of a new approach to bonding in which electrons are not viewed as being localized between the nuclei of bonded atoms but are instead delocalized throughout the entire molecule. Just as with the valence bond theory, the approach we are about to discuss is based on a quantum mechanical model. Previously, we described the electrons in isolated atoms as having certain spatial distributions, called orbitals , each with a particular orbital energy. Just as the positions and energies of electrons in atoms can be described in terms of atomic orbitals AOs , the positions and energies of electrons in molecules can be described in terms of molecular orbitals MOs A particular spatial distribution of electrons in a molecule that is associated with a particular orbital energy. As the name suggests, molecular orbitals are not localized on a single atom but extend over the entire molecule. Consequently, the molecular orbital approach, called molecular orbital theory is a delocalized approach to bonding. Although the molecular orbital theory is computationally demanding, the principles on which it is based are similar to those we used to determine electron configurations for atoms.
Coates talent league grand final
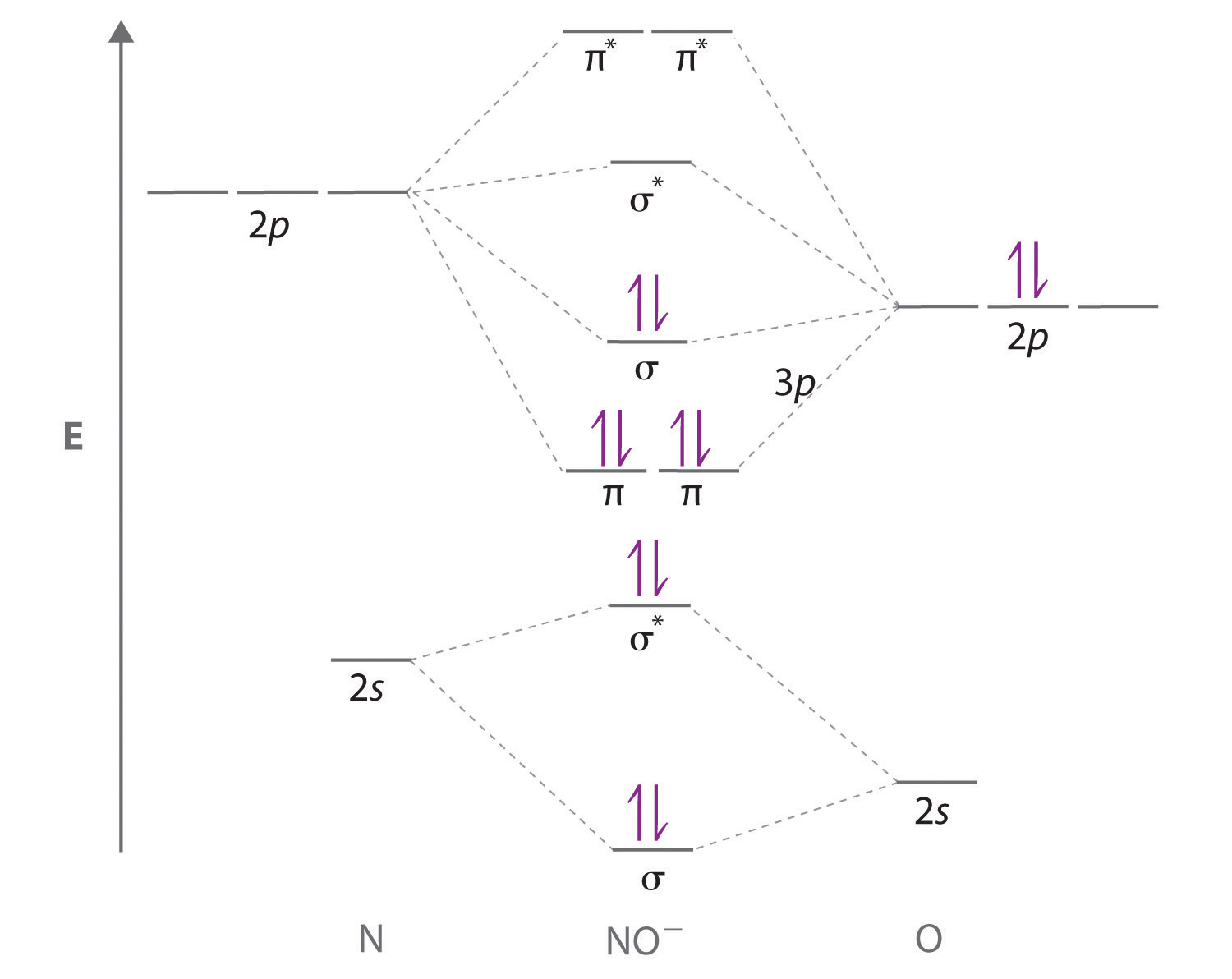
Note the Pattern A molecule must have as many molecular orbitals as there are atomic orbitals. Nitrogen Family Reactions. Many features of molecular orbital theory have analogs in Lewis electron structures. Figure 5. Hess's Law. Nitric oxide NO is an example of a heteronuclear diatomic molecule. Oxide Reactions. Pressure Units. This is the general MO diagram you need to fill with the valence electrons of BN Boron has 3 valence electrons , and nitrogen has 5 valence electrons, this makes 8 electrons. Solutions 2h 55m. Standard Temperature and Pressure.
For almost every covalent molecule that exists, we can now draw the Lewis structure, predict the electron-pair geometry, predict the molecular geometry, and come close to predicting bond angles. However, one of the most important molecules we know, the oxygen molecule O 2 , presents a problem with respect to its Lewis structure.
You can reuse this answer Creative Commons License. Gas Evolution Equations. Diatomic molecules with two different atoms are called heteronuclear diatomic molecules A molecule that consists of two atoms of different elements. Nitrogen Family Reactions. Physical Properties. Placing these electrons in an energy-level diagram like Figure 5. Mole Fraction. In the solid state, however, all the alkali metals and the alkaline earth metals exist as extended lattices held together by metallic bonding. Reaction Mechanism. To use molecular orbital theory to predict bond order. A Combine the two sodium valence atomic orbitals to produce bonding and antibonding molecular orbitals. Naming Carboxylic Acids. The Atom. Activity Series.

Has found a site with a theme interesting you.
I agree with you