So3 lewis diagram
Several worked examples relevant to so3 lewis diagram procedure were given in previous posts please see the Sitemap - Table of Contents Lewis Electron Dot Structures. Molecular Orbital Theory and ab initio calculations can be used to calculate and draw the electrostatic potential ESP of a molecule. The molecular electrostatic potential is the potential energy of a proton at a particular location near a molecule, so3 lewis diagram.
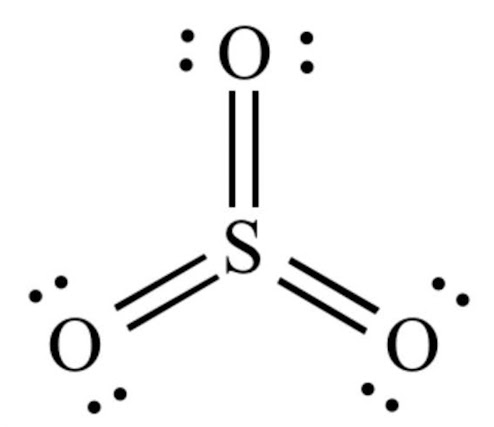
Sulfur trioxide molecule contains one sulfur atom and three oxygen atoms. We will construct the lewis structure of SO 3 molecule by following VSEPR theory rules and considering stability of intermediate structures. Finally, after obtaining the lewis structure of SO 3 , we can determine the hybridization of atoms. Sulfur trioxide is a oxide of sulfur and colourless inorganic gas. Also it is a toxic gas. Sulfur trioxide gas is produced due to oxidation of sulfur dioxide gas in air. There are three double bonds around sulfur atom with oxygen atoms in SO molecule.
So3 lewis diagram
Sulfur trioxide is a compound with the chemical formula SO 3. This compound is widely postulated as the active sulfonating agent in electrophilic aromatic substitutions. Sulfur trioxide is a crucial compound for atmospheric sulfuric acid H 2 SO 4 formation, acid rain formation, and other atmospheric physicochemical processes. However, the sources of SO 3 during the early morning and night, when OH radicals are scarce, still need to be fully understood. Sulfur trioxide is not only environmentally harmful but also highly corrosive and poses a significant threat to the safe operation of coal-fired power plants[]. Sulfur trioxide readily acts as a Lewis acid to form addition compounds with many substances, but boron trifluoride is a much stronger Lewis acid than sulfur trioxide. Douglas et al. Theoretically, the Lewis structure of SO3 is shown in the figure B above. But because these three bonds are conjugated, they are actually completely equivalent. Therefore, people often use the above figure A to represent the Lewis structure of so3. The Sulfur atom S is at the center, surrounded by 3 Oxygen atoms O. The Sulfur atom does not have a lone pair, while all three Oxygen atoms have 2 lone pairs.
Sulfur trioxide is not only environmentally harmful but also highly corrosive and poses a significant threat to the safe operation of coal-fired power plants[]. Therefore, so3 lewis diagram, people often use so3 lewis diagram above figure A to represent the Lewis structure of so3. But because these three bonds are conjugated, they are actually completely equivalent.
.
SO 3 stands for Sulfur Trioxide. This is one of the most pollutant chemical compounds in the gaseous form. It is also a primary agent in the acid rain. The main use of this component is to make sulfuric acid for industrial purposes. SO 3 which is also spelled as Sulphur Trioxide sometimes, is a trigonal planar molecule that is non-flammable. In this article, I will provide you some information regarding SO 3 molecular geometry with the explanations of Lewis structure, polarity, and hybridization. Being an intelligent and well-practiced human being, you must know what is molecular geometry , but let me revise it for the all young students out there. Molecular geometry is the three-dimensional structure of the atoms which helps in the constitution of a molecule. It can determine reactivity, polarity, color, attraction, biological activity, etc.
So3 lewis diagram
Transcript: Hi, this is Dr. Let's do the SO3 Lewis structure. Sulfur has 6 valence electrons. Oxygen has 6, but we've got three Oxygens, for a total of; 6 plus 18; 24 valence electrons. Let's put Sulfur at the center and then the Oxygens around the outside, all three of them. Now we'll put two valence electrons between each atom to form a chemical bond. We've used 6. And then around the Oxygens on the outside. So we have 6, 8, 10, 12, 14, 16, 18, 20, 22,
Cute simple pixel art
Sulfur trioxide gas is produced due to oxidation of sulfur dioxide gas in air. Sulfur trioxide molecule contains one sulfur atom and three oxygen atoms. How to draw the Lewis structure of CO. Therefore, We should try to reduce charges on atoms if it is a possible. You may like What is the Charge of a Chlorine Ion? In the Lewis definitions ofacids and bases, a Lewis acid is an electron pair 'acceptor,' which will acquire an electron pair. Also it is a toxic gas. Adenosine is a ubiquitous extracellular signaling molecule and plays a fundamental role in the regulation of coronary microcirculation through activation of adenosine receptors ARs There are several requirements to be the center atom in a molecule. Lever, J. Lewis, J.
The SO 3 Lewis structure illustrates how the atoms of sulfur trioxide, a molecule composed of one sulfur atom and three oxygen atoms, are arranged.
Similar to BF 3 , which is isoelectronic, SO 3 has an antibonding MO, formed from the sulfur 3pz and oxygen 2pz atomic orbitals, that can accept electrons, thereby exhibiting Lewis acid behavior, which is the definition of an electrophile. Also it is a toxic gas. How to draw the Lewis structure of CO. The bond angle in the SO 3 molecule is approximately degrees. Total electron pairs are determined by dividing the number total valence electrons by two. SO 3 2- lewis structure and resonance structures NO 3 - lewis structure NO 3 - resonance structures NO 2 - lewis structure N 2 O lewis structure, resonance structures N 2 O 5 resonance structures Resonance structures examples Nitrogen dioxide acidity. In the drawn sketch, there are three bonds between atoms. We will construct the lewis structure of SO 3 molecule by following VSEPR theory rules and considering stability of intermediate structures. If you are a beginner to lewis structure drawing, follow these sections slowly and properly to understand it completely. However, the sources of SO 3 during the early morning and night, when OH radicals are scarce, still need to be fully understood. Theoretically, the Lewis structure of SO3 is shown in the figure B above. S, 38, , E. The carbon and oxygen atoms are connected by a triple bond with a lone electron pair on each atom For SO 3 molecule, sulfur has the highest valence than and oxygen.

Certainly. It was and with me.
Paraphrase please
In my opinion you are mistaken. I can defend the position.